The race for a COVID-19 vaccine is the most urgent challenge the world’s medical research community has faced during our lifetime.
Rather than a short-distance sprint, however, this race is more akin to a long-distance obstacle course with many hurdles to clear before we have a vaccine approved for general use.
In the meantime, people everywhere will have to get used to some degree of social distancing and other restrictions on our daily life.

On the bright side, the race for a COVID-19 vaccine is heating up and many significant milestones have already been achieved.
This article will cover:
- A new normal until we have a COVID-19 vaccine
- Who is winning the race for a COVID-19 vaccine
- When will a vaccine be available to the general public
- Stages a vaccine must pass through before it is available for general use
A Vaccine is Essential if We are to Return to Normal
In late April and early May many countries began easing up on the very tight lockdowns they began to implement in March. These looser restrictions, however, are still far from what we are accustomed to as ‘normal’.
Political leaders, health officials and scientists have been explaining that we will not be returning to complete ‘normalcy’ until a COVID-19 vaccine is available for general use:
- On April 6th, Dr. Anthony Fauci, a key member of the US government’s coronavirus task force stated “If ‘back to normal’ means acting like there never was a coronavirus problem, I don’t think that’s going to happen until we do have a situation where you can completely protect the population.”, “Ultimately, the show stopper will obviously be a vaccine, where you can vaccinate people and you won’t have it.”
- On April 5th, Bill Gates, co-chair of the the Bill & Melinda Gates Foundation which has already committed $250 million to the fight against COVID-19 stated “Things will not get back to truly normal until we have a vaccine that has gotten out to, basically, the entire world”.
- On April 9th, Canada’s Prime Minister Justin Trudeau stated “Normality as it was before will not come back full-on until we get a vaccine for this… That will be a very long way off”.
- On April 12th, Dr. David Nabarro, a special envoy of the WHO on the coronavirus stated “We think it’s going to be a virus that stalks the human race for a quite a long time to come, until we can all have a vaccine that will protect us”.
- On April 13th, while discussing the risk of countries loosening restrictions too soon, Tedros Adhanom Ghebreyesus, director-general of the World Health Organization (WHO) stated “Ultimately, the development and delivery of a safe and effective vaccine will be needed to fully interrupt transmission”.
- On April 13th, New York state Governor Andrew Cuomo stated “I don’t think ultimate resolution comes until you have a vaccine. That’s 18 months.”
- On April 15th, United Nations secretary-general Antonio Guterres said ”A safe and effective vaccine may be the only tool that can return the world to a sense of ‘normalcy’, saving millions of lives and countless trillions of dollars”.
- On April 14th, even as China had basically controlled its original COVID-19 outbreak, respiratory scientist Zhong Nanshan who heads a government task-force to contain the COVID-19 outbreak in China stated that the coronavirus was still a serious threat and we would need to rely on a vaccine. Mr. Zhong had earlier stated that there is no guarantee people who survive COVID-19 once would become immune for life. So counting on herd immunity is not an option and vaccines will still be the key to ultimately tackling the virus.
The Race for a COVID-19 Vaccine Begins
The starting gun was fired to start the race for a COVID-19 vaccine on January 10th, 2020.
Just one week prior, Chinese scientists had isolated the novel coronavirus from one of the earliest patients in Wuhan.
On Friday, January 10th they presented the genetic sequence for SARS-CoV-2, the official name of the COVID-19 coronavirus, to the world community by posting it on a public database.
This is much quicker than it took to get the genetic sequence for SARS, a disease caused by another coronavirus which caused havoc back in 2002/2003.
Dr. Peter Hotez, a vaccine scientist at Baylor College of Medicine in Houston stated “With SARS, it took almost a year to be able to identify and map the full genetic code. Now we’re doing this in just a few weeks.”
Previously, labs needed to obtain a sample of the live virus and be able to grow it before vaccine development work could begin. Innovations in vaccine development in recent years, however, have resulted in new ways to develop vaccines based on the virus’s genetic sequence.
Participants Join the Race for a COVID-19 Vaccine
In early February the China Association for Vaccines announced 17 of its members were already working on COVID-19 vaccines.
A list built by the Coalition for Epidemic Preparedness Innovations (CEPI), a global public-private alliance which is funding several COVID-19 vaccine development programs, includes 115 COVID-19 vaccine candidates at varying stages of development as of April 8th. The advanced programs come from 19 different countries.
A World Health Organization (WHO) list titled “Draft landscape of COVID 19 candidate vaccines”, last updated on May 5th, includes 108 vaccine candidates of which 8 have already entered human clinical trials.
This sounds like more than enough programs to ensure success, but Jerome Kim, director-general of the International Vaccine Institute (IVI) warns that vaccine development is characterized by a high failure rate of “often 93%” between initial development through to when a vaccine has been approved for use by the general public.
Who is Winning the Race for a COVID-19 Vaccine
If we are to comment on who is winning the race for a COVID-19 vaccine we would have to say it is CanSino Biologics Inc., a Chinese biopharmaceutical company.
At least one could make this argument at the beginning of May 2020.
CanSino was founded by Yu Xuefeng, who did post-graduate studies and then worked for many years in Canada; hence the ‘Can’ in CanSino.
CanSino’s vaccine candidate was developed in collaboration with a team led by Dr. Chen Wei a major general in the People’s Liberation Army (PLA), China’s military. She is also a noted virologist who heads the Institute of Biotechnology at the Academy of Military Medical Sciences.
CanSino’s vaccine candidate began phase-one human trials on March 19th in Wuhan city, where the novel coronavirus epidemic broke out.
CanSino completed the phase-one trial within just a few weeks and began phase-two clinical trials on April 12th, 2020.
It is the first COVID-19 vaccine candidate in the world to enter this stage of the long vaccine development and approval process.
CanSino’s phase-two trials are scheduled to be completed by January 31, 2021.
Moderna – First Human Trial of a COVID-19 Vaccine
Another contender for the lead in the race for a COVID-19 vaccine, and the one which has received the most press in western countries, is the US company Moderna Inc.
Moderna’s vaccine candidate was developed in cooperation with the US government’s National Institute of Allergy and Infectious Diseases (NIAID), headed by Dr. Anthony Fauci.
The stock price of Moderna surged 28 percent on February 24th when it was reported that it had delivered the ‘first coronavirus vaccine’ to the US National Institutes of Health (NIH), which the NIAID is a part of.
In fact, what Moderna had delivered to the NIH was just a vaccine candidate, a stage to which many COVID-19 vaccine programs had also achieved by this time.
One such program, launched by a Chinese company from Chengdu, Clover Biopharmaceuticals, announced two weeks earlier, on February 10th, that its COVID-19 vaccine candidate had already successfully detected antigen-specific antibody.
Its website states “Clover is the first company in the world to disclose a 2019-nCoV vaccine candidate that can successfully be recognized by antibodies produced by previously-infected patients”.
Moderna’s COVID-19 vaccine candidate, however, was the first in the world to be injected into a human.
The Moderna phase-one clinical trial began on March 16th, just a few days before CanSino’s phase-one launch. It is being carried out at the Kaiser Permanente Washington Health Research Institute in Seattle, Washington, USA with 45 volunteers.
On May 1st, Moderna’s CEO Stephane Bancel said that the phase-one trial safety data “looks positive”.
The company has submitted its phase-two application to the FDA which it expects to begin in May or June 2020. Phase-three would then commence in the fall of 2020.
Another US Contender – Inovio
The third COVID-19 vaccine candidate to enter human trials was one developed by Inovio of the US. Its phase-one clinical trial was launched on April 6th.
Inovio expects the results from its phase-one trial to be available in June, after which it would quickly move into phase-two trials.
In January, as the race for a COVID-19 vaccine had just begun, Inovio CEO J. Joseph Kim was relishing the competition against Moderna, stating “We’re starting at the exact same time and this is a great opportunity for us to go mano a mano with Moderna”, “I like our chances.”
First Inactivated Vaccine To Begin Human Trials
Another first for China in the race for a COVID-19 vaccine was achieved by a program launched by the China National Biotech Group Co Ltd. (CNBG), a subsidiary of the Chinese pharmaceutical giant Sinopharm (also known as the China National Pharmaceutical Group Co.). This program is a collaboration with the Wuhan Institute of Biological Products Co and the Wuhan Institute of Virology.
The vaccine candidate developed by this collaboration was the the first Inactivated vaccine in the world to begin human trials, just after receiving approval on April 12th.
On April 25th, just 12 days after beginning phase-one trials, Sinopharm launched the phase-two trial for this vaccine.
Different Types of Vaccines
An Inactivated vaccine is based on a long established development approach that has produced many successful vaccines commonly in use today, including the annual flu shot.
Although requiring a longer design and testing process, the likelihood of success for this type of vaccine could be higher than the types of vaccine CanSino, Moderna and Inovio are working on.
Cansino is developing a Viral Vector vaccine.
Moderna and Inovio are developing their vaccines based on even more novel approaches. Both are categorized in the broader category of Nucleic Acid vaccines, with Moderna developing a mRNA vaccine and Inovio developing a DNA vaccine. Although these vaccines are quick to create, there are as yet no Nucleic Acid vaccines approved for use in humans.
Other COVID-19 vaccine development programs are also employing new innovations. The University of Queensland in Australia is using its proprietary Molecular Clamp technology. Canada’s Medicago is using a plant-based platform to grow Virus-like particles (VLPs) for its antigen. And Shanghai’s own Stemirna Therapeutics is employing its patented core shell structure platform to produce its mRNA vaccine.
For an introduction to these novel technologies and different types of vaccines please see our developing the COVID-19 vaccine article.
Different vaccines can induce varying degrees of immunity in different population groups. So the more types of vaccines our scientists can successfully develop to prevent COVID-19, the better.
In mid-February China announced its research institutes and companies were working on 5 different types of COVID-19 vaccines including inactivated vaccine, recombinant vaccine, live vector vaccine, nucleic acid-based vaccine and live attenuated vaccine.
In early March when Zheng Zhongwei, an official at China’s National Health Commission stated “the development progress for vaccines in China is similar or somewhat ahead of those in foreign countries”, he credited “The reason for the rapid progress of vaccine research (in China) is all research institutions have worked together to accelerate research regardless of cost.”
The UK Moves into Third Place
On Thursday April 23rd, The University of Oxford launched human trials for its ChAdOx1 nCoV-19 vaccine, another candidate developed using the viral vector approach.
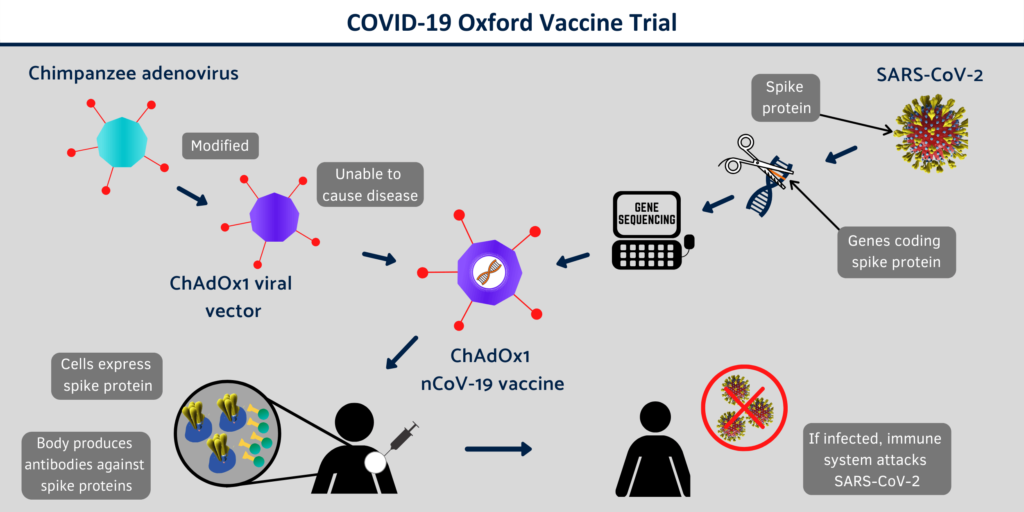
The University of Oxford is combining their phase-one and phase-two trials by enrolling over 500 volunteers at once and scheduling the two-phase trial to to run for the next 6 months.
Unlike a typical single phase phase-one trial, Oxford is hoping to also test for efficacy in protection from COVID-19, and not just safety, right from the start.
The British health secretary Matt Hancock announced the launch of the University of Oxford trial and committed financial support to both it and another program at the Imperial College London (an mRNA candidate), stating that the government will ‘throw everything at it.”; i.e. the search for a COVID-19 vaccine.
Germany Tied for Third
On April 23rd it seemed that the UK had moved solely into third place of the national standings in the race for a COVID-19 vaccine. However, on April 29th, it was reported that Germany’s BioNTech had also begun human trials for its mRNA vaccine the same day as Oxford University.
BioNTech is receiving financial support from the pharmaceutical giant Pfizer and China’s Fosun Pharma to develop their vaccine.
Pfizer said it hoped to manufacture millions of doses of the vaccine by the end of 2020, and hundred’s of millions of doses in 2021.
More COVID-19 Vaccine Candidates Enter Human Trials
On April 13th, the National Medical Products Administration (NMPA) of China approved another Inactivated vaccine candidate developed by Sinovac to enter human trials. Human trials commenced shortly after on April 16th with 144 volunteers.
Meng Weining, Sinovac’s senior director for overseas regulatory affairs, says the company hopes to start phase-two trials by mid-May and have results by the end of June.
Other COVID-19 Vaccine Candidates Approved for Human Trials
On April 8th Novavax, a US company announced its Subunit vaccine candidate was shown to be highly immunogenic in animal models and that it would begin human trials in mid-May.
On April 27th, Sinopharm’s second Inactivated vaccine candidate was approved for phase-two trials. This candidate was developed by another of Sinopharm’s subsidiary’s; Beijing Institute of Biological Products Co Ltd., in cooperation with the Chinese Center for Disease Control and Prevention.
On April 28th it was reported that Linear Clinical Research, a clinic in Australia has begun to recruit volunteers for human trials to test Clover Biopharmaceuticals’ S-Trimer vaccine candidate.

When will a Vaccine be Available to the General Public
The first widely publicized forecast for the availability of a COVID-19 vaccine for general use was stated by the WHO’s director-general Dr. Tedros Adhanom Ghebreyesus on February 11th. Dr. Tedros said “The first vaccine could be ready in 18 months”, i.e. the summer of 2021.
The timeframe of 12-18 months has since been the standard narrative used by most health officials.
On March 12th, when urging patience despite rapid progress with the Moderna vaccine candidate, Dr. Fauci stated “It’s going to take a year to a year-and-a-half to really know if it works.”
On April 14th, two months after Dr. Tedros’s statement, WHO spokeswoman Dr. Margaret Harris re-emphasized “We really shouldn’t be expecting to see the vaccine at least for 12 months or longer.”
Eighteen months sounds like a long time to wait, especially as we are in the midst of a pandemic. Often, however, new vaccines take 5-10 years of development and testing before they receive regulatory approval.
In November 2019 the first vaccine for Ebola, which was designed at Canada’s National Microbiology Laboratory in 2005, was finally approved for general use.
Writing in The Economist, Bill Gates states “My hope is that, by the second half of 2021, facilities around the world will be manufacturing a vaccine. If that’s the case, it will be a history-making achievement: the fastest humankind has ever gone from recognising a new disease to immunising against it.”
Don’t Count on a COVID-19 Vaccine being Ready in Eighteen Months
Many voices caution that counting on a COVID-19 vaccine to be ready within even eighteen months is way too optimistic.
In an article titled “Sober Up! 25 Reasons Not to Count on COVID Vaccine for Herd Immunity in 1-2 years,” SVB Leerink analyst Geoffrey Porges forecasts that it will take several years rather than months before we can administer a vaccine to enough of the world’s population to control COVID-19.
Porges writes “We view the current expectations for a vaccine in this timeframe as the equivalent of standing 24 feet (the usual distance is 8 feet) from a dartboard, with one dart in hand, and counting on a bullseye from one throw. It is theoretically possible, but highly unlikely, that such expectations are correct.”
Porges has a degree in medicine and worked for Merck in their vaccine division for several years.
Global analytics firm Clarivate, using an analytical tool it developed gives the Moderna candidate just a 5% chance of success. Inovio’s vaccine candidate received a slightly higher 15% chance of success.
Even if they prove to be successful, Clarivate estimates both will require over 5 years before they are approved.
Considering that neither Moderna nor Inovio have developed a licensed vaccine of any kind, this skepticism is understandable.
Optimistic Forecasts
There have been some more optimistic voices; forecasting a COVID-19 vaccine will get to the finish line even earlier than 12 months.
US President Donald Trump is one. On March 2nd he stated “I’ve heard very quick numbers, that of months. And I’ve heard pretty much a year would be an outside number. So I think that’s not a bad range. But if you’re talking about three to four months in a couple of cases, a year in other cases”.
Although his words immediately met with pushback from Dr. Fauci he is not alone in this optimistic forecast.
On April 12th, the president of the European Commission Ursula von der Leyen said she is hopeful that a COVID-19 vaccine will be ready by the end of 2020.
One of China’s leading vaccine experts, Wu Zunyou, chief epidemiologist of the Chinese Center for Disease Control and Prevention outlined a theoretical timeline of six months for a Chinese vaccine candidate to go through all three phases of clinical trials.
Based on current progress for COVID-19 vaccines in China and Mr. Wu’s best case scenario, we could see a vaccine candidate completing all 3 phases of clinical trials in China by September or October 2020.
Mr. Wu Zunyou recently gave an enlightening interview in English about China’s battle with COVID-19.
Dr. Fauci has also begun to show more optimism. In an interview with The Associated Press on April 14th he said it might be possible to finish phase-three trials on Moderna’s vaccine candidate quicker than his original forecast, possibly by “mid to late winter of next season (the 2020/2021 winter).”
But, he emphasized “Please let me say this caveat: That is assuming that it’s effective. See, that’s the big ‘if,’ It’s got to be effective and it’s got to be safe.”
Vaccine Developers Optimistic Forecasts
The leaders of several COVID-19 vaccine development programs are also expressing confidence they can have a vaccine ready long before the summer of 2021.
The team at the University of Oxford has set a target of this autumn to be able to mass produce their vaccine.
Sarah Gilbert, who leads the team states that she is “80% confident” the vaccine would work, and could be ready by September.
The Oxford vaccine project’s official website states “The best-case scenario is that by the autumn of 2020 we could have an efficacy result from the phase III trial to show that the vaccine protects against the virus, alongside the ability to manufacture large amounts of the vaccine, but these best-case timeframes are highly ambitious and subject to change.”
A team of researchers at the University of Bern is hoping to be able to inoculate the entire Swiss population in October with the vaccine it is developing. It is already in discussion with pharmaceutical giants including Novartis for manufacturing its vaccine candidate in large enough quantities.
The lead of the Swiss vaccine development team, Dr Martin Bachmann, says that they will begin to produce for the world market after they have immunized “a lot of the Swiss population in the next six months.”
Operation Warp Speed
On April 29th, the US administration launched Operation Warp Speed, a project to speed up the availability of a COVID-19 vaccine.
The aim of the project is to have 300 million doses of a vaccine manufactured and available to Americans by January 2021.
On discussing this new effort at the end of April, Dr. Fauci said he now thinks this goal is attainable.
Stages in the Race for a COVID-19 Vaccine
The race for a vaccine will generally go through the following 7 stages commencing from when a new contagious disease appears through until when we have a vaccine available for general use.
Below, we also look at more milestones achieved and issues yet to be tackled in the race for a COVID-19 vaccine.
First Stage – Identifying the Virus
As mentioned above, Chinese scientists isolated the virus from patients in early January 2020 and presented the genetic sequence of Sars-CoV-2 to the international scientific community on January 10th.
Second Stage – Vaccine Candidate
Several research institutes and biopharmaceutical companies around the world started work on developing vaccine candidates for COVID-19 within hours of the SARS-CoV-2 genetic sequence being published that Friday night.
CEO J. Joseph Kim of Inovio stated “Our team worked around the clock and was able to design a spike-focused vaccine by that Sunday night,” just two days after receiving the genome sequence.
US government and military scientists also began working on a vaccine candidate immediately.
Dr Gordon Joyce, a scientist working for the US Walter Reed Army Institute of Research stated “On 10 January, when the first sequence became available for the new coronavirus, we started working straight away. At that stage we’d heard it was pretty bad in China but we didn’t know the full extent”.
Obtaining a Live Virus
Some programs employing traditional approaches to develop the COVID-19 vaccine still required a live virus. Chinese labs had access to the live virus in early January, but a live virus can not be easily transported to labs in other countries.
Australia’s Peter Doherty Institute has been credited as the first laboratory outside of China to grow the SARS-CoV-2 virus. They accomplished this on January 28th, 2020.
They have since shared the virus with some Australian laboratories and the WHO. Likewise, however, it is not easy to share the virus with labs in other countries.
The US Centers for Disease Control and Prevention (CDC) quickly isolated a virus from one of the first US patients who tested positive on January 20th, and made it broadly available to the US research community.
The Walter Reed Army Institute quickly began using the CDC provided virus to work on a second type of vaccine in their labs.
Japan’s National Institute of Infectious Diseases succeeded in isolating a SARS-CoV-2 virus on January 31st, 2020 and began their vaccine development work immediately.
In mid-March it was still considered a great breakthrough when a team of scientists from Canada’s Sunnybrook research institute, McMaster University, and the University of Toronto were able to isolate SARS-CoV-2 and grow it in their secure containment facility.
Third Stage – Preclinical Studies on Animals
Before testing on humans, preclinical studies are conducted to see if the vaccine is safe and can stimulate an immune response. Usually a number of different species of animals are used for testing at this stage.
Moderna Skips Animal Trials
Although it was announced on February 25th that Moderna’s vaccine candidate had achieved an immune reaction in mice it has since been reported that Moderna and the NIAID skipped much of the standard preclinical animal testing phase and went straight into human trials.
Some experts consider this a big gamble as well as a safety risk, but others feel it is necessary. At a meeting of scientists representing government-funded research organizations and drugmakers around the world convened by the WHO in mid-February it was agreed that the threat from COVID-19 was so great that vaccine candidates could begin human trials without the requirement for completing animal trials first.
First Vaccine Candidates to Begin Comprehensive Animal Testing
On April 2nd the two vaccine candidates developed by Inovio and the University of Oxford were the first to be submitted for comprehensive preclinical trials of a vaccine with an animal model.
The animal trials for the Inovio and Oxford University candidates are being carried out at the Commonwealth Scientific and Industrial Research Organisation (CSIRO) in Australia and will include ferrets.
Testing on ferrets is seen as much more indicative of what the immune response of humans will be to the vaccine than mice.
Ferrets have the same ACE2 receptors on cells in their lungs as humans. The SARS-CoV-2 virus uses the ACE2 to break into cells leading to humans contracting COVID-19.
The results of the CSIRO trials, therefore, will still be of great interest despite several human trials having already begun. These results are expected by June.
Sinovac Inactivated Vaccine Animal Trials
It is not clear what animal tests were conducted for the CanSino vaccine candidate, but data has been published for the Sinovac Inactivated vaccine tests on mice, rats and rhesus macaques.
Rhesus macaques are a type of monkey which show COVID-19 symptoms similar to humans.
The test results published revealed that after receiving the vaccine the monkeys became highly immune to COVID-19.
The antibodies the vaccine induced in mice and rats were also tested in vitro against several strains of the virus isolated from patients from different countries. The antibodies were able to neutralize the various strains, indicating that the virus has not mutated enough to affect the efficacy of the Sinovac vaccine.
The Sinovac tests also indicate that no antibody dependent enhancement (ADE) was caused by the vaccine. ADE is when the body reacts worse to a virus after receiving a vaccine.
This phenomenon occurred with some of the SARS vaccine candidates in 2003 as well as with some other animal coronavirus vaccines; so it is a big concern.
Although not peer-reviewed, and receiving some skepticism, many scientists agree the Sinovac data results are very encouraging.
Oxford University’s Animal Trials
Like Sinovac, Oxford University also tested its vaccine candidate on rhesus macaque monkeys.
After being vaccinated, the monkey’s were then exposed to large amounts of the SARS-CoV-2 virus. Almost one month after exposure the monkey’s had still not contracted COVID-19.
First Peer-Reviewed Study of a COVID-19 Vaccine
The University of Pittsburgh is credited with publishing the first peer reviewed animal trial study for its vaccine candidate.
The tests on mice showed the PITTCov protein Subunit vaccine produced high levels of effective SARS-CoV-2 antibodies.
Fourth Stage – Phase-one Clinical Trials on Humans
Phase-one trials on humans are focused on testing for safety of the vaccine. The vaccine is injected into a small number of volunteers who are not at risk of catching the disease. Usually between 40 and 100 volunteers are recruited for this stage.
Although CanSino’s trial took less than a month to complete, most phase-one trials take about 3 months. So by late summer 2020 we could have the results from many different COVID-19 phase-one vaccine trials.
Reactions in Phase-one Trials
CanSino’s phase-one trial showed some adverse effects in volunteers. It was reported that some volunteers had fevers “with the high-dose group having more people with a fever 38.5 degrees Celsius.” The fevers usually broke within 24 hours.”
Moderna’s phase-one trials have yet to report any negative side effects, except for a soreness in the arm where the injection was given.
Fifth Stage – Phase-two Clinical Trials
In phase-two clinical trials safety is still a key concern. However, scientists now begin to focus on efficacy, i.e. if the vaccine can spur an effective immune response in humans.
After receiving the vaccine, scientists will frequently check blood samples from the volunteers to see if they have produced antibodies effective in the prevention of contracting COVID-19.
For this phase several hundred volunteers are recruited and separated into different groups. One group will receive a placebo, while others will receive varying numbers of doses and dosages for the purpose of designing vaccination procedures.
Sixth Stage – Phase-three Clinical Trials
If phase one and two trials show that a vaccine candidate is both safe and effective, testing on a much larger group of volunteers can begin.
In this phase thousands if not tens of thousands of volunteers will be used to gather a huge amount of scientific data from different demographic groups over a longer period of time.
Scientists want to see how effective the vaccine is when the volunteers are at risk of exposure to the virus in natural disease conditions.
Phase-three trials generally take 6-8 months, if not longer, to complete.
Testing in Real-world Conditions – Area of Spread
Phase-three trials are more effective and quicker to conclude when carried out in an area of rapid disease spread.
Vaccine candidates developed in 2003 to prevent SARS stopped research at this stage because its virus, SARS-CoV, was no longer spreading in any region.
For SARS, we were fortunate that the virus did not return, but most experts now feel that even if this first outbreak of COVID-19 is controlled, SARS-CoV-2 is likely to be still with us and could cause new waves, just like the annual flu.
So, we should certainly not abandon the race for a COVID-19 vaccine even if the situation becomes much improved!
Human Challenge Trials
If the disease is no longer spreading rapidly in any region, one option to speed up development would be to carry out human challenge trials.
Healthy young individuals who would likely only get a mild case of COVID-19 if infected would be used as test subjects. They would first be vaccinated and then intentionally exposed to the wild virus to see if the infection can be prevented.
Such trials are rare and somewhat dangerous, but have been used throughout the history of vaccine development.
Already, potential volunteers are being recruited and scientists are pushing the US FDA to consider this option.
Immediate Mass Deployment of a Vaccine
If COVID-19 is still spreading rapidly, opposite to considering human challenge trials would be to consider if a vaccine candidate that enters phase-three trials should be made available to the general public rather than just a controlled number of volunteers.
Zeng Yixin, deputy director of China’s National Health Commission, explained that scientists in China can apply for emergency approval to deploy vaccines still being tested into emergency use by the public if necessary.
Two highly rated German COVID-19 vaccine developers, BioNTech and CureVac, have called for the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) to waive the requirement for rigorous Phase-three trials so they can have hundreds of millions of doses of their vaccines available by the end of 2020.
Dr. Mike Ryan, executive director of the WHO’s emergencies program, however, urged caution. He explained that there is only one thing more dangerous than a virus “and that’s a bad vaccine. We have to be very, very, very careful in developing any product that we’re going to inject into potentially most of the world’s population”.
China’s Wang Junzhi, an academician with the Chinese Academy of Engineering, also urged caution, “Even during an emergency, we cannot lower the standards of safety and efficacy when reviewing a vaccine. The public is paying a lot of attention.”
Limited Deployment
Another possible strategy would be to allow healthcare workers and the immune compromised such as the elderly and those with pre-existing medical conditions to use the vaccine before it becomes available to the general public.
The WHO estimates there are 45 million health workers on the frontlines tackling COVID-19 around the globe.
On March 20th Moderna’s CEO Stephane Bancel said although their mRNA vaccine, “is not likely” to be available to the general public for at least 12 to 18 months, “it is possible that under emergency use, a vaccine could be available to some people, possible including healthcare professionals, in the fall of 2020.”
On April 23rd, Chinese Center for Disease Control and Prevention (CDC) Director Gao Fu said that China could have a vaccine for emergency use in September, which could be applied on particular groups, such as medical staff. And it could be administered more widely in early 2021.
Sinopharms’ Inactivated vaccine has already been registered for emergency production and use according to Chinese laws.
Johnson & Johnson (J&J), who are preparing manufacturing capacity for more than one billion doses of their vaccine, claims they could could have the first batches available for emergency use in early 2021.
J&J is producing a fascinating video series on the road to a COVID-19 vaccine.
Seventh Stage – Mass Production and Deployment to the General Public
Normally, only after a vaccine candidate has proven effective and is, at least, close to being licensed do pharmaceutical companies begin to manufacture it in volume.
Since the number of doses of the COVID-19 vaccine that will be required to beat this pandemic is so large, a huge amount of new production capacity will first need to be built. Even after the capacity is ready, it will take considerable time to manufacture and distribute the billions of doses that will be required to protect everyone on earth.
One way to accelerate the availability of a vaccine to everyone is for pharmaceutical companies to ‘proceed at risk’, i.e. build the capacity for manufacturing the COVID-19 vaccine and begin production before it has received regulatory approval.
